
Moseley bombarded a number of chemical elements with x rays and observed the pattern formed by the reflected rays. The concept of atomic number evolved from the historic research of Henry Gwyn-Jeffreys Moseley in the 1910s. Accordingly the atomic number is often omitted from a nuclear symbol, as in 16O, where the superscript represents the atomic mass (a attribute than does vary with isotopes of an element). In nuclear chemistry, an element ’s atomic number is written to the left and below the element ’s symbol The number of protons for a particular element never varies, if one changes the number of protons one is changing the element. It is always the smaller whole number found in association with an element ’s symbol in the table. The atomic number of an element can be read directly from any periodic table. Since each proton carries a single positive charge, the atomic number is also equal to the total positive charge of the atomic nucleus of an element. Oxygen ’s atomic number is, therefore, eight. For example, the nucleus of an oxygen atom contains eight protons and eight neutrons. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.The atomic number of an element is equal to the number of protons in the nucleus of its atom. 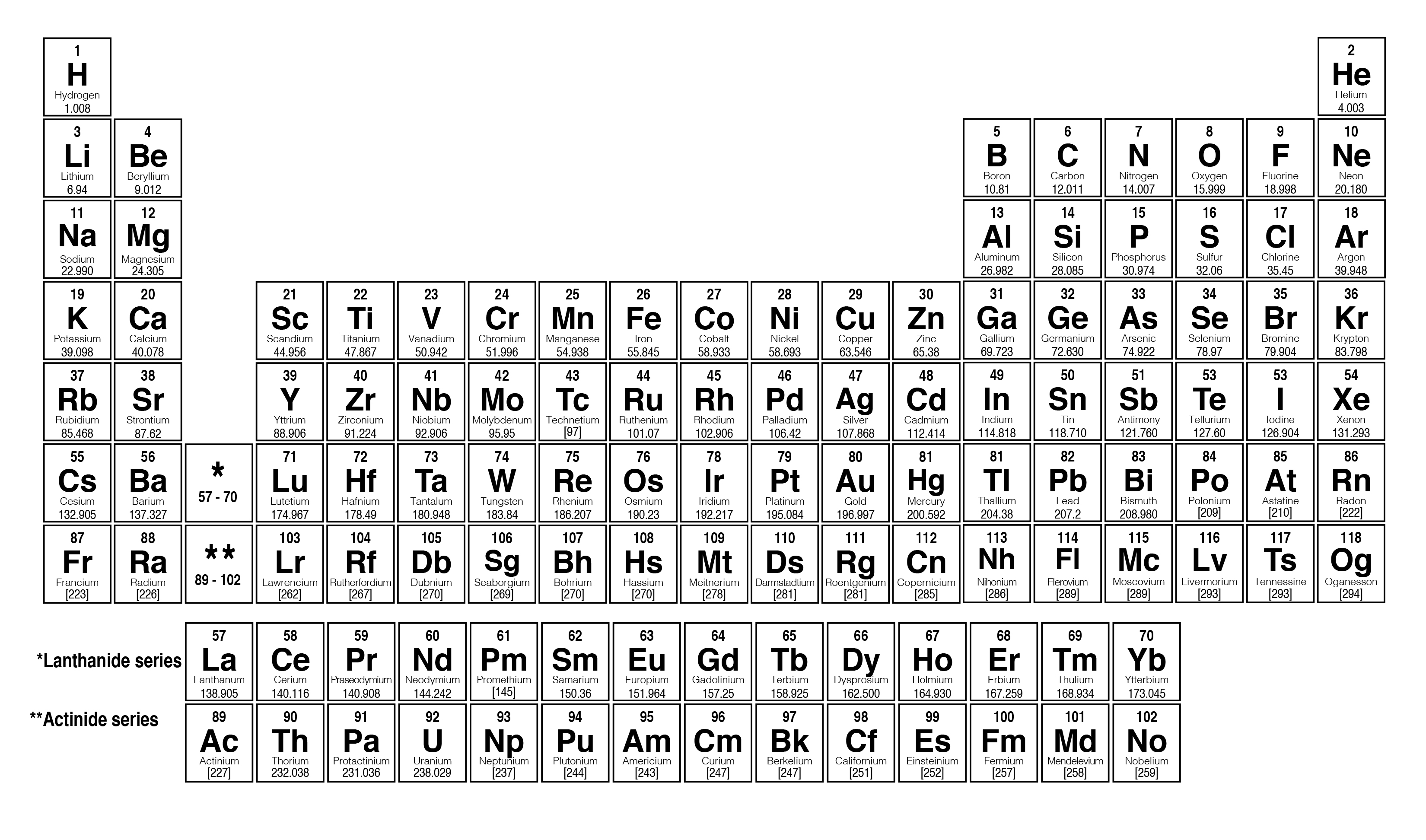
Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). 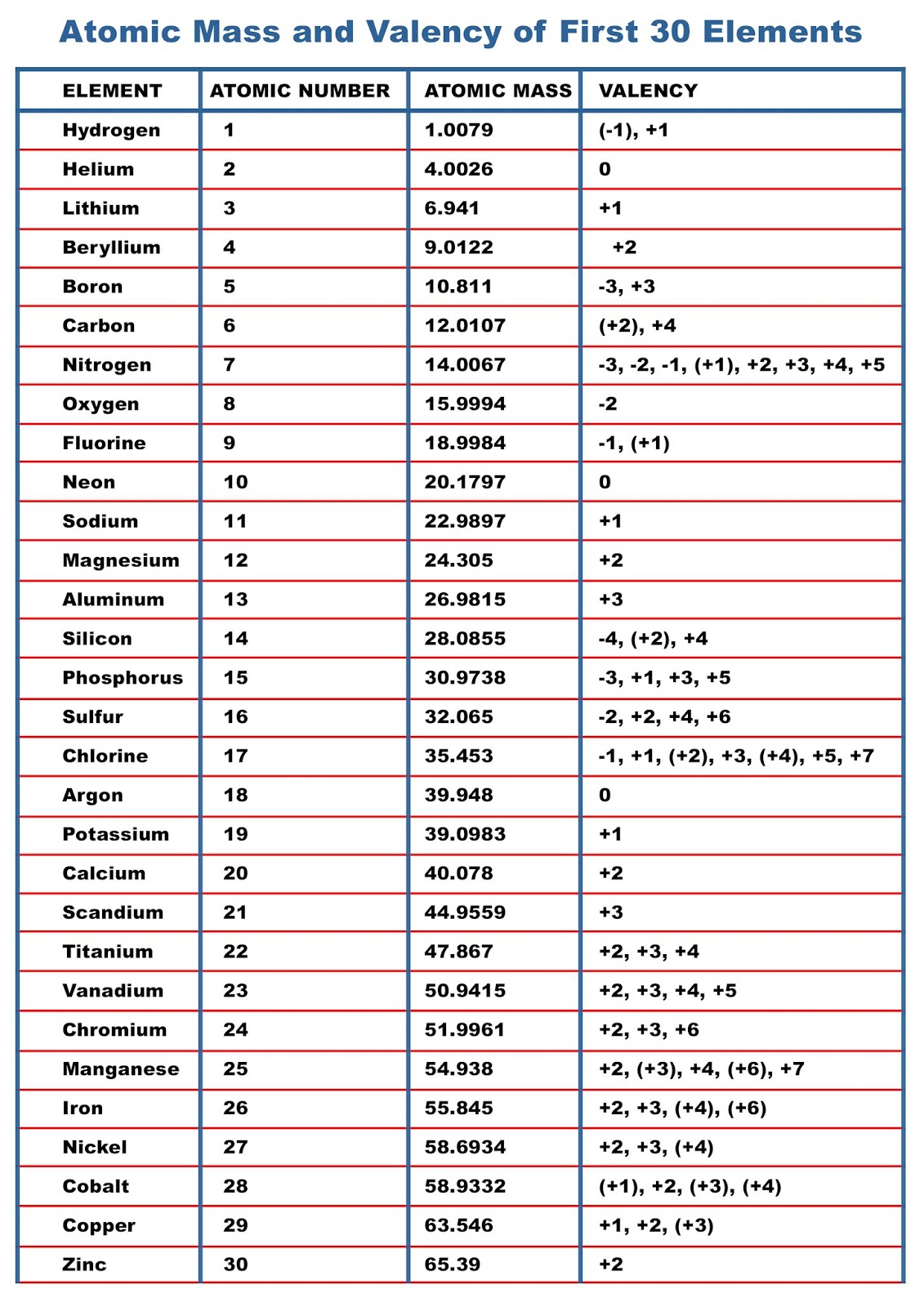
Electronic configuration of first 30 elementsįree Gift for you: Interactive Periodic Table Valence electrons of first 30 elementsĢ). If you have any queries, feel free to comment below.Īlso let me know, has this article helped you or not?ġ). I hope you have got the complete list of 118 elements and their symbols and atomic number along with their atomic mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
